In January, the Food and Drug Administration took the bold step of reclassifying surgical mesh as a class III device. This is a recognition by the FDA that surgical mesh is a high-risk device and that great care should be taken in determining whether it is appropriate for the device to be used in the treatment of pelvic organ prolapse (POP).
A History of Surgical Mesh
Surgical mesh has a long history of use in the medical field. The product was initially used in the 1950’s to treat hernias, and it is still used for that purpose today. Surgical mesh works by providing temporary support for organs so that they are able to heal. In the 1970’s, doctors started using mesh to treat pelvic organ prolapse (POP). This type of prolapse often happens to women after childbirth, and it can cause many complications. By the 1990’s gynecologists began implanting mesh transvaginally in order alleviate prolapse symptoms.
In 2002, the FDA started approving mesh devices specifically for transvaginal use. The agency initially classified the device as class II. This rating meant that use of the mesh carried moderate risk of complications. Since this original classification, however, the FDA has seen a high level of reported adverse events associated with transvaginal mesh.
Mesh Products Cause Complication when used Transvaginally
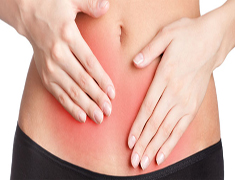
As long as doctors have been using surgical mesh transvaginally, women have been having complications. Some of the side effects women have suffered include vaginal bleeding, pain during intercourse, perforations of the bowel, and urinary problems. Due to the many reported issues with the devices, the FDA has released a number of warnings. These warnings include: a safety communication in 2008, a warning to doctors and consumers in 2011, and an order to manufacturers in 2012 to conduct postmarket surveillance studies on the effectiveness and safety of transvaginal mesh products. These warnings culminated in the decision last month to reclassify transvaginal mesh as a class III device.
Class III devices are considered high-risk, and are treated differently because of this classification. The FDA order requires manufacturers to look into safety concerns by complying with a rigorous premarket approval application. It also puts Doctors and patients on notice that transvaginal mesh can be very harmful, and it should only be used after thorough analysis of the patient’s situation.
We Can Help
If you or a loved one have been harmed by a mesh device, you may be entitled to compensation. Our attorneys have extensive experience in dealing with these types of cases, and we can help you navigate the confusing world of tort law. Give us a call today.
1-877-542-4646




 According to an
According to an