Granuflo and NaturaLyte (dialysis injury)

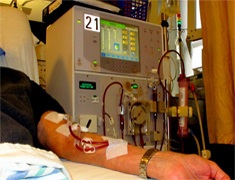
Dialysis Lawyers McSweeney / Langevin Investigating Injuries and Deaths from GranuFlo and NaturaLyte
Fresenius Medical Care products, GranuFlo and NaturaLyte, are chemicals used in dialysis machines to help clean blood. Granuflo and NaturaLyte contain a substance called bicarbonate. Bicarbonate is used to prevent a buildup of acid in the blood. However, GranuFlo and NaturaLyte may contain more bicarbonate than rival products. An overdose of bicarbonate can lead to cardiac arrest and possibly death.
Many victims received Granuflo and NaturaLyte at DaVita or Fresenius dialysis. Together, the two companies control roughly two-thirds of the dialysis market in the United States.
Risks / Side Effects of GranuFlo and NaturaLyte
- Hemodialysis Cardiopulmonary Arrest
- Cardiopulmonary Death
- Heart problems
- Metabolic alkalosis
- Low blood pressure
- Myocardial infarction (MI)
- Heart attack
- Sudden chest pain
- Sudden heart attack
- Nausea
- Sweating
- Stroke
- Death
These symptoms and side effects will usually occur during dialysis or shortly thereafter.
German Based Fresenius Medical Misled the Public
Fresenius Medical conducted an internal study of GranuFlo and NaturaLyte. The results were not good. The internal study revealed an up to eight-fold increase in the risk of cardiopulmonary arrest and sudden cardiac death. This information was detailed in a November 4, 2011 Internal Memo sent to Fresenius medical directors and physicians. The internal memo warned a small number of doctors of the sharp increase in instances of sudden death from cardiac arrest. The internal memo states “[t]his issue needs to be addressed urgently.”
However, Fresenius did not address the issue “urgently.” In fact, Fresenius failed to immediately inform the Food and Drug Administration of dangers associated with GranuFlo and NaturaLyte. Fresenius only warned the public of the safety risks and dangers of GranuFlo and NaturaLyte when an individual sent an anonymous copy of the internal memo to the FDA.
FDA Recall of Fresenius Medical Care’s GranuFlo Concentrate and NaturaLyte Liquid
The FDA has classified the Granuflo and NaturaLyte recall as a Class I recall because of the potential risk of serious injury or death.
Fresenius GranuFlo and NaturaLyte Timeline / News
11/04/2011 Fresenius Medical Care Internal Memo warning certain doctors of the sharp increase in death following use of GranuFlo and NaturaLyte
03/29/2012 FDA Class I Recall of GranuFlo and NaturaLyte
03/29/2012 Fresenius Medical Care issues an urgent product notification letter concerning GanuFlo and NaturaLyte
06/14/2012 The New York Times published an article concerning Fresenius’ failure to warn of the risks associated with GranuFlo and NaturaLyte
11/07/2012 News Release
03/29/2013 News Release and video regarding Granuflo and NaturaLyte
04/14/2014 FDA Recalls Fresenius Medical’s NaturaLyte due to dangerous levels of bacteria

1-877-542-4646
FREE Case Review
Free Confidential Case Evaluation
To contact us for a free review of your potential case, please fill out the form below or call us toll free 24 hrs/day by dialing 1-877-542-4646
An asterisk (*) indicates a required field.